EN
-
Home
Home
-
Product
Product
-
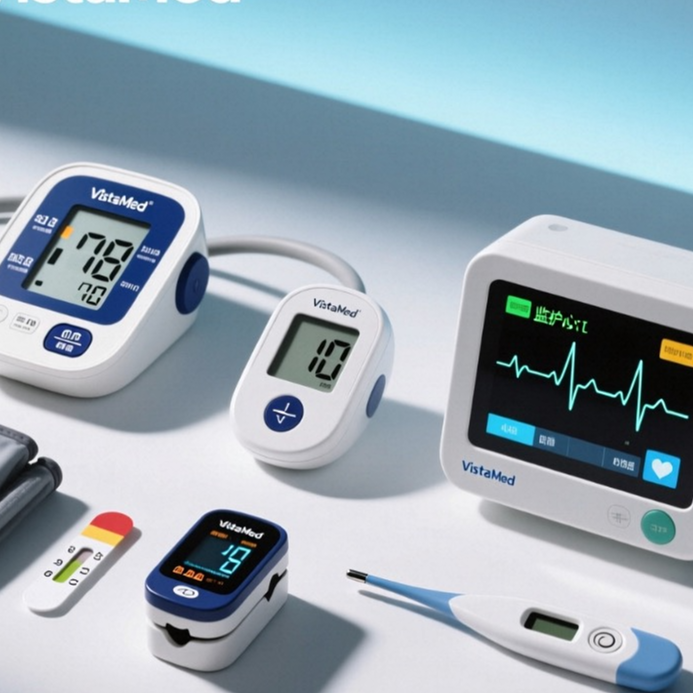
ECG & Heart Monitor
ECG & Heart Monitor
-
Blood Glucose Monitor
Blood Glucose Monitor
-
Blood Pressure Monitor
Blood Pressure Monitor
-
Pulse Oximeter
Pulse Oximeter
-
Digital Thermometer
Digital Thermometer
-
ECG & Heart Monitor
- Solutions
- About us
- Service
-
News
News