Unwavering Support for Healthcare Providers
2026/02/25

2026/02/21
Author: Dr. Evelyn Reed, MD
Lead Medical Content Reviewer & Clinical Advisor at VistaMed Technologies
As a licensed physician specializing in medical communications, Dr. Reed translates complex clinical data and technical information into clear, accurate, and actionable insights for healthcare professionals and partners.
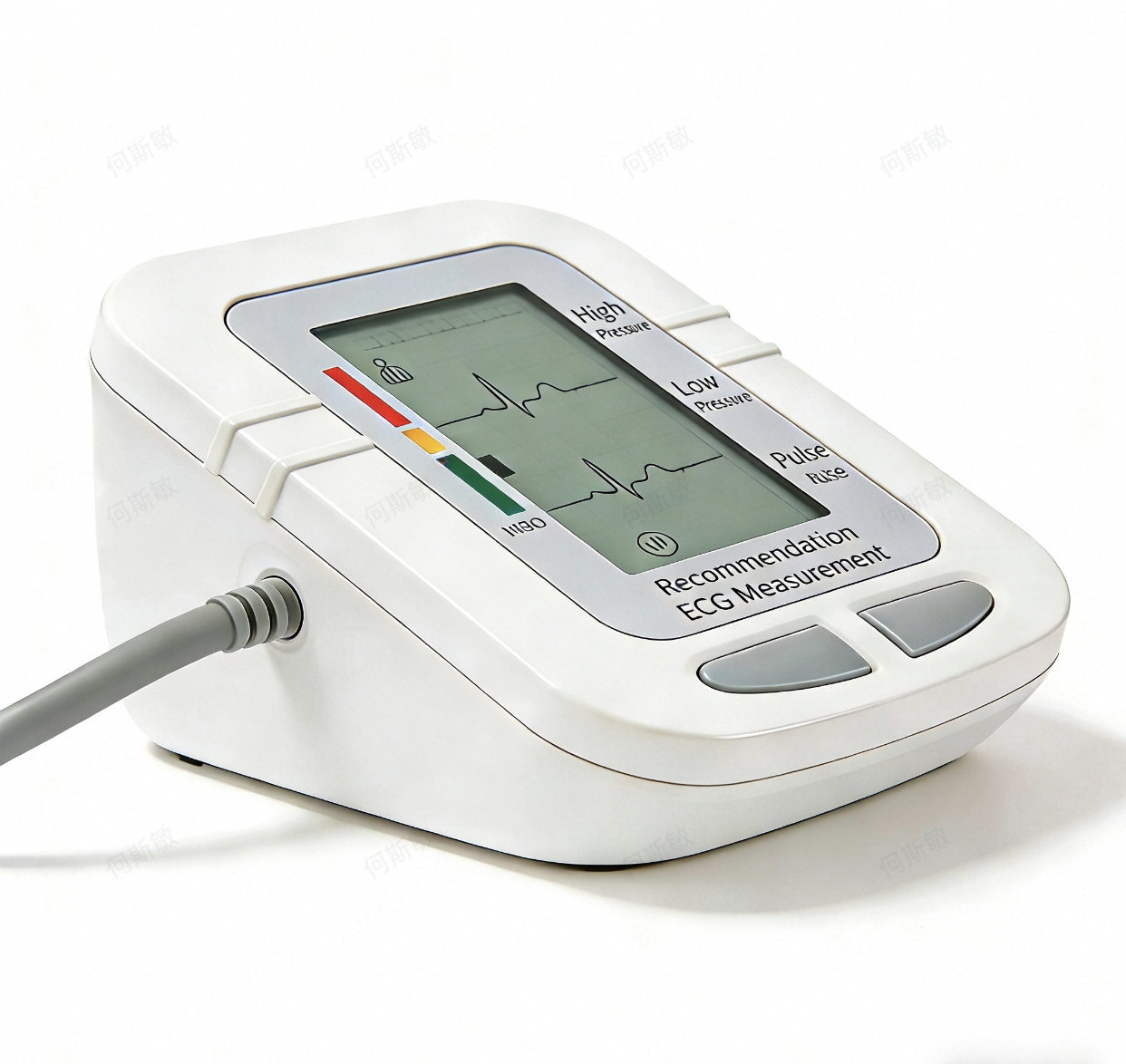
In my work reviewing clinical data, I once came across a report of a well-meaning salesperson who told a patient that the jagged line on their new single-lead ECG monitor "looked like AFib." The patient, understandably panicked, went to the emergency room, only to be told it was just a noisy, artifact-filled tracing from a low-quality device.
That salesperson made a critical error. They crossed the line from being a product expert to providing a medical interpretation—a practice that is both inappropriate and a massive liability. For you, the medical device distributor, knowing where that line is and how to talk about it is crucial for your business. This FAQ is designed to give you the evidence-based answers you need.
This is the single most dangerous myth a distributor can believe. The idea that your role includes helping a customer, whether a clinician or a patient, interpret a single-lead ECG is incorrect and exposes your business to significant risk.
From a clinical and legal perspective, your job is not to interpret the ECG; your job is to provide a professional-grade tool that generates a high-fidelity, artifact-free signal that a qualified clinician can trust for their own interpretation. Selling a device means selling the quality of its data output. The interpretation of that data belongs exclusively to a licensed healthcare provider.
You should have a clear, immediate, and consistent response: “I cannot provide a medical interpretation, as I am not a qualified clinician. This device is a screening tool designed to capture a high-quality heart rhythm for a doctor to review. You must share this result with your physician for a proper diagnosis.”
This response does three things:
You can then pivot the conversation to what you can talk about: the quality of the signal the device produces, which is the foundation of any accurate interpretation by a clinician.
You prove it by talking about the engineering and regulatory rigor behind the device's software. The tracing itself is only half the product; the algorithm that cleans and presents that signal is the other half.
This is where you can demonstrate deep expertise. Explain that the software in a professional-grade device is considered Software as a Medical Device (SaMD). This means it is subject to intense regulatory scrutiny. You can reference the risk-based classification frameworks developed by global bodies like the International Medical Device Regulators Forum (IMDRF) to show that your manufacturer understands these complexities.
A quality manufacturer designs their algorithm to minimize artifact and produce a clean, stable baseline, making it easier for a clinician to identify P waves, QRS complexes, and T waves. You are selling signal clarity, not a diagnosis.
The risks are immense, especially concerning data security. A simple consumer ECG gadget might send an unencrypted email of the tracing. A professional system is designed with security as a core feature.
This is a critical differentiator. The US FDA has a strong focus on medical device cybersecurity, and a manufacturer who can't speak intelligently about their threat modeling and data encryption is not a serious player. Highlighting that your product, like our SmartBP-Connect with its ECG option, uses a HIPAA-compliant app and secure cloud infrastructure is a powerful sales argument for a clinical customer.
Furthermore, a professional system comes from a partner committed to data integrity. Our commitment to clinical validation is demonstrated through collaborations, such as a remote monitoring study with the Cardiovascular Research Institute at Stanford University. A partner trusted by world-class researchers is a partner you can trust with your customers' data.
You should highlight the features that enable a clinician to make a confident interpretation.
SmartBP-Connect, you can highlight the ability to correlate the ECG rhythm strip with a validated blood pressure reading taken at the same time, providing the clinician with a richer dataset for their assessment.
Your goal is to sell a tool that makes a clinician's job easier and more effective.
"As a distributor, your greatest asset is the trust of your clinical customers. When it comes to ECGs, that trust is built on your ability to provide a device that delivers a clean, reliable signal. Leave the interpretation to the clinicians, and focus on providing them with the highest quality data possible. That is the cornerstone of a responsible—and profitable—business."
— Dr. Evelyn Reed, MD
About the Author
Dr. Evelyn Reed, MD, serves as Lead Medical Content Reviewer & Clinical Advisor at VistaMed Technologies. With over a decade of experience in medical communications, she specializes in translating complex clinical data and technical information into clear, accurate, and actionable insights for healthcare professionals. This article draws on her deep experience evaluating the clinical evidence and real-world performance of vital signs monitoring technologies.
Clinically & Regulatory Reviewed By: Dr. Michael Bauer, PhD, Head of Clinical Research
Medical Disclaimer:The information provided is for informational purposes and intended for a B2B audience of healthcare professionals and procurement decision-makers. It is not a substitute for professional medical or financial advice. TCO and ROI results may vary based on facility size, usage patterns, and local market conditions. All certifications and regulatory clearances referenced are accurate as of the date of publication. Please contact VistaMed Technologies for the most current documentation.